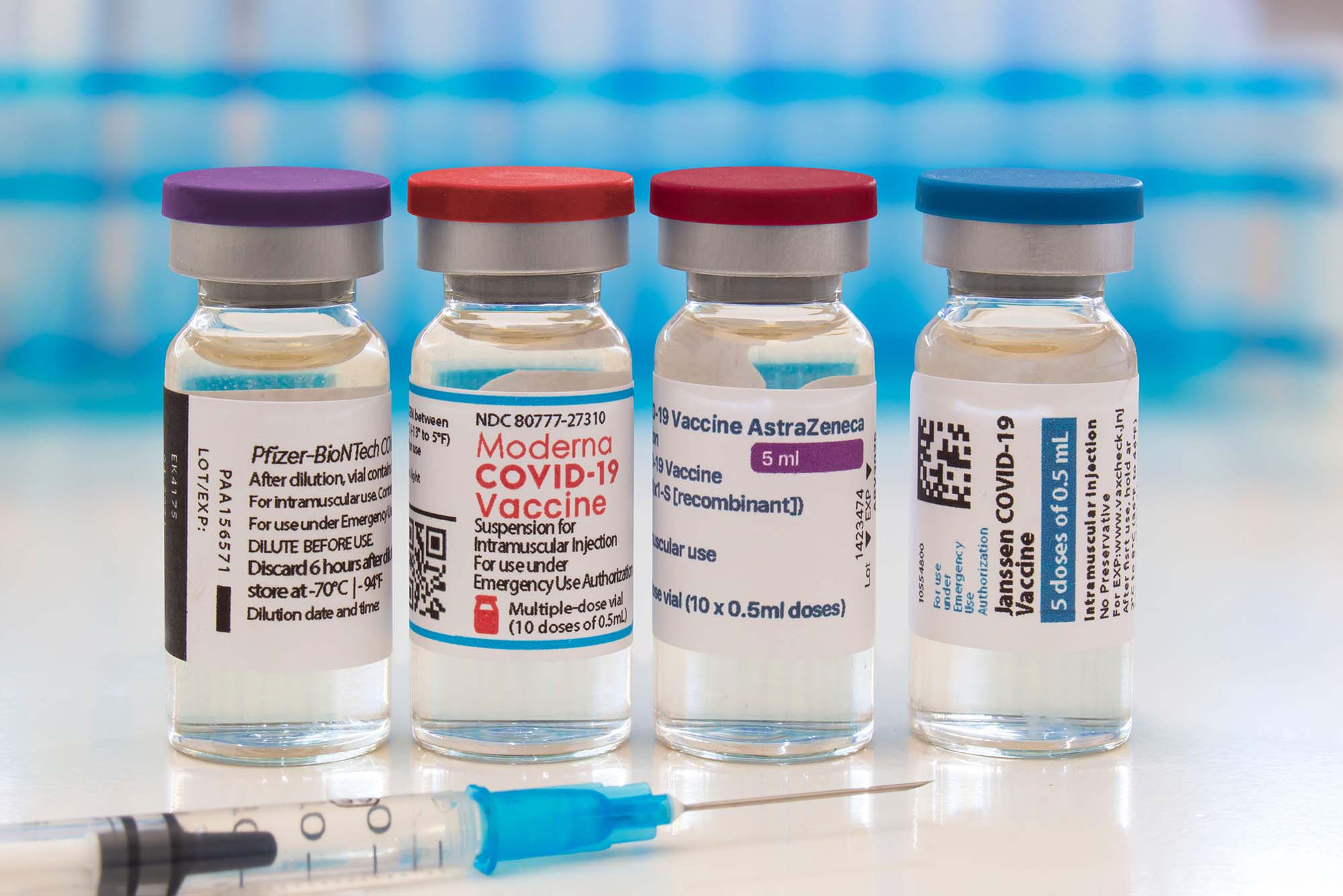
FDA authorizes another booster dose of the Pfizer or Moderna COVID-19 vaccine for people age 50 and up - The Boston Globe

FDA, CDC, Western States authorize booster doses of the Pfizer COVID-19 vaccine for 16- and 17-year-olds : Oregon Health News Blog

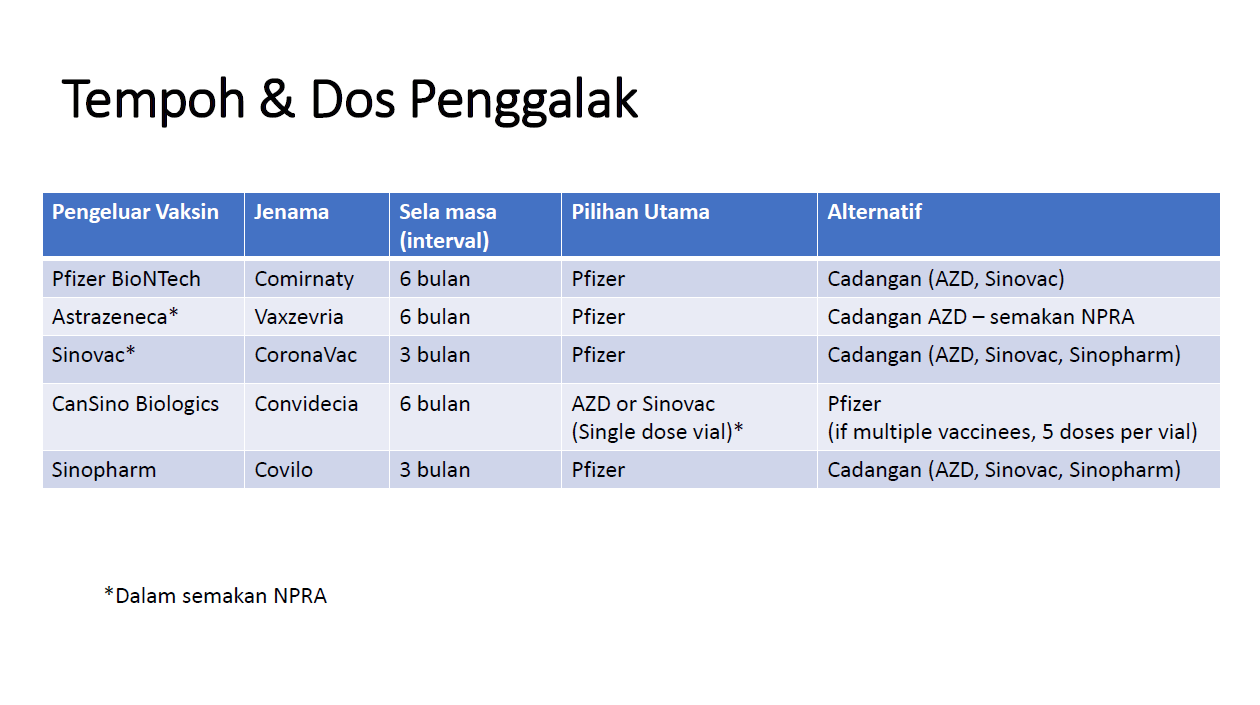
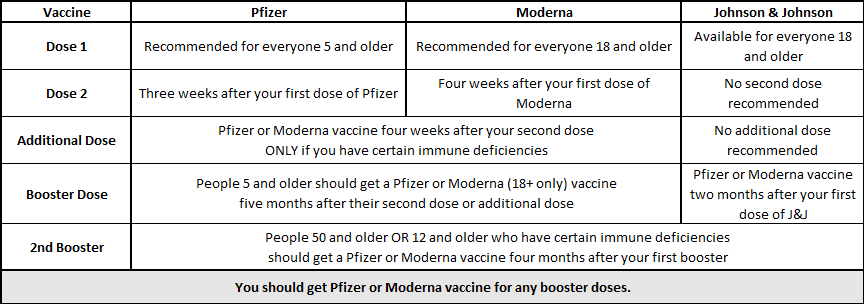
Coronavirus (COVID-19) Update: FDA Shortens Interval for Booster Dose of Moderna COVID-19 Vaccine to Five Months | FDA

Frederick County Health Department - Are you eligible for a Pfizer booster dose of COVID-19? Yes if you got Pfizer doses 1 & 2, your 2nd dose was more than 6 months

Covid-19, Fda estende l'idoneità per la dose booster del vaccino Pfizer-BioNTech ai bambini da 5 a 11 anni - AboutPharma

Governor Michelle Lujan Grisham - Pfizer COVID-19 vaccine booster doses are now available. If you received both doses of the Pfizer vaccine at least six months ago, you may be eligible at
LVHN Prepared to Give Pfizer COVID-19 Vaccine Booster Shot to Seniors, At-Risk Workers, Vulnerable Populations Starting Sept. 27