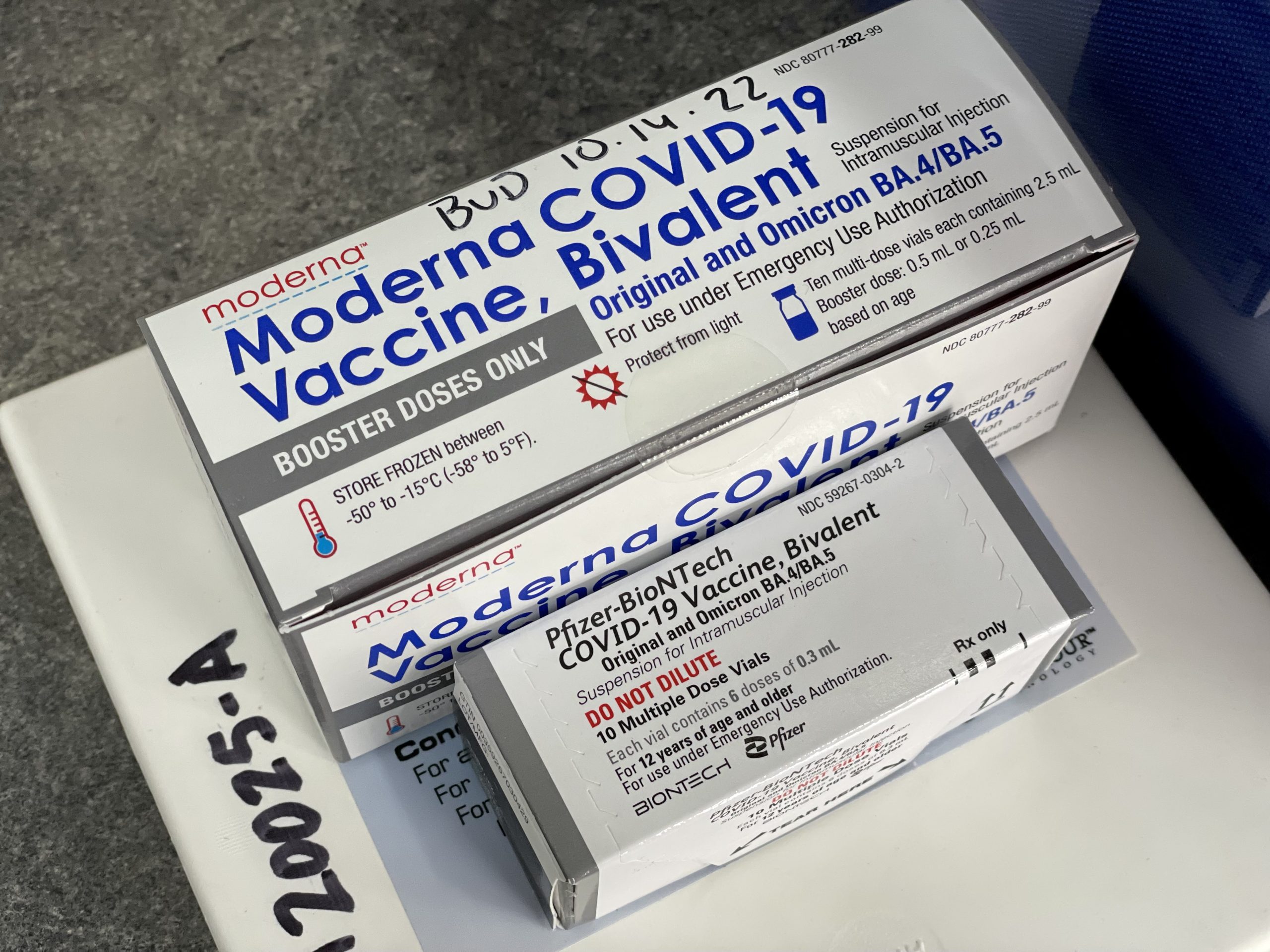
Omicron-Updated COVID-19 Boosters and $20 Gift Cards at CCPH - Cow Creek Band of Umpqua Tribe of Indians

Vaccines | Free Full-Text | Vaccine Confidence and Uptake of the Omicron Bivalent Booster in Tennessee: Implications for Vulnerable Populations

Early Estimates of Bivalent mRNA Booster Dose Vaccine Effectiveness in Preventing Symptomatic SARS-CoV-2 Infection Attributable to Omicron BA.5– and XBB/XBB.1.5–Related Sublineages Among Immunocompetent Adults — Increasing Community Access to Testing ...

Effect of hybrid immunity and bivalent booster vaccination on omicron sublineage neutralisation - The Lancet Infectious Diseases

Immunogenicity of bivalent omicron (BA.1) booster vaccination after different priming regimens in health-care workers in the Netherlands (SWITCH ON): results from the direct boost group of an open-label, multicentre, randomised controlled trial -

Updated COVID-19 booster targets subvariants responsible for 98% of cases today | Malheur County Health Department
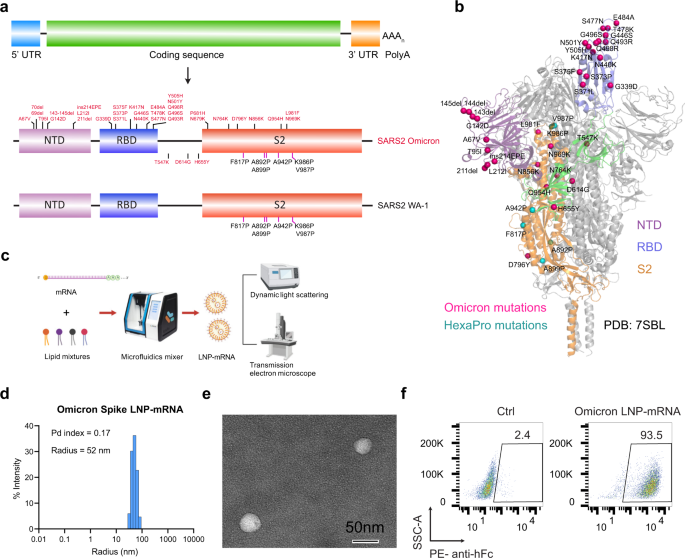
Omicron-specific mRNA vaccination alone and as a heterologous booster against SARS-CoV-2 | Nature Communications

Low neutralization of SARS-CoV-2 Omicron BA.2.75.2, BQ.1.1 and XBB.1 by parental mRNA vaccine or a BA.5 bivalent booster | Nature Medicine