MenACWY-CRM conjugate vaccine booster dose given 4–6 years after priming: Results from a phase IIIb, multicenter, open label study in adolescents and adults - ScienceDirect
![Safety Profile | MENVEO [Meningococcal (Groups A, C, Y and W-135) Oligosaccharide Diphtheria CRM₁₉₇ Conjugate Vaccine] Safety Profile | MENVEO [Meningococcal (Groups A, C, Y and W-135) Oligosaccharide Diphtheria CRM₁₉₇ Conjugate Vaccine]](https://www.menveohcp.com/content/dam/cf-pharma/hcp-menveo/en_US/safety/Safety_chart_Mobile.png)
Safety Profile | MENVEO [Meningococcal (Groups A, C, Y and W-135) Oligosaccharide Diphtheria CRM₁₉₇ Conjugate Vaccine]
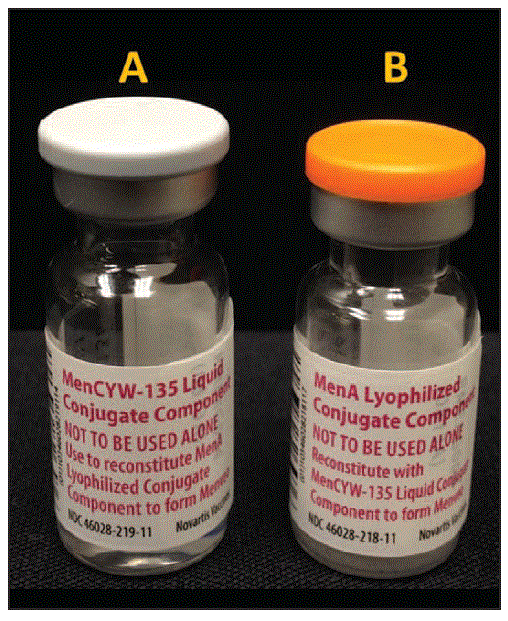
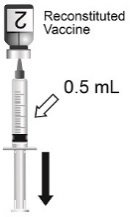
Notes from the Field: Administration Error Involving a Meningococcal Conjugate Vaccine — United States, March 1, 2010–September 22, 2015 | MMWR