Biological Compatibility of Medical Service (ISO 10993) - Superlab CRO -health food, medical device, chemicals, pharmaceutical research (GLP&TAF accredited)

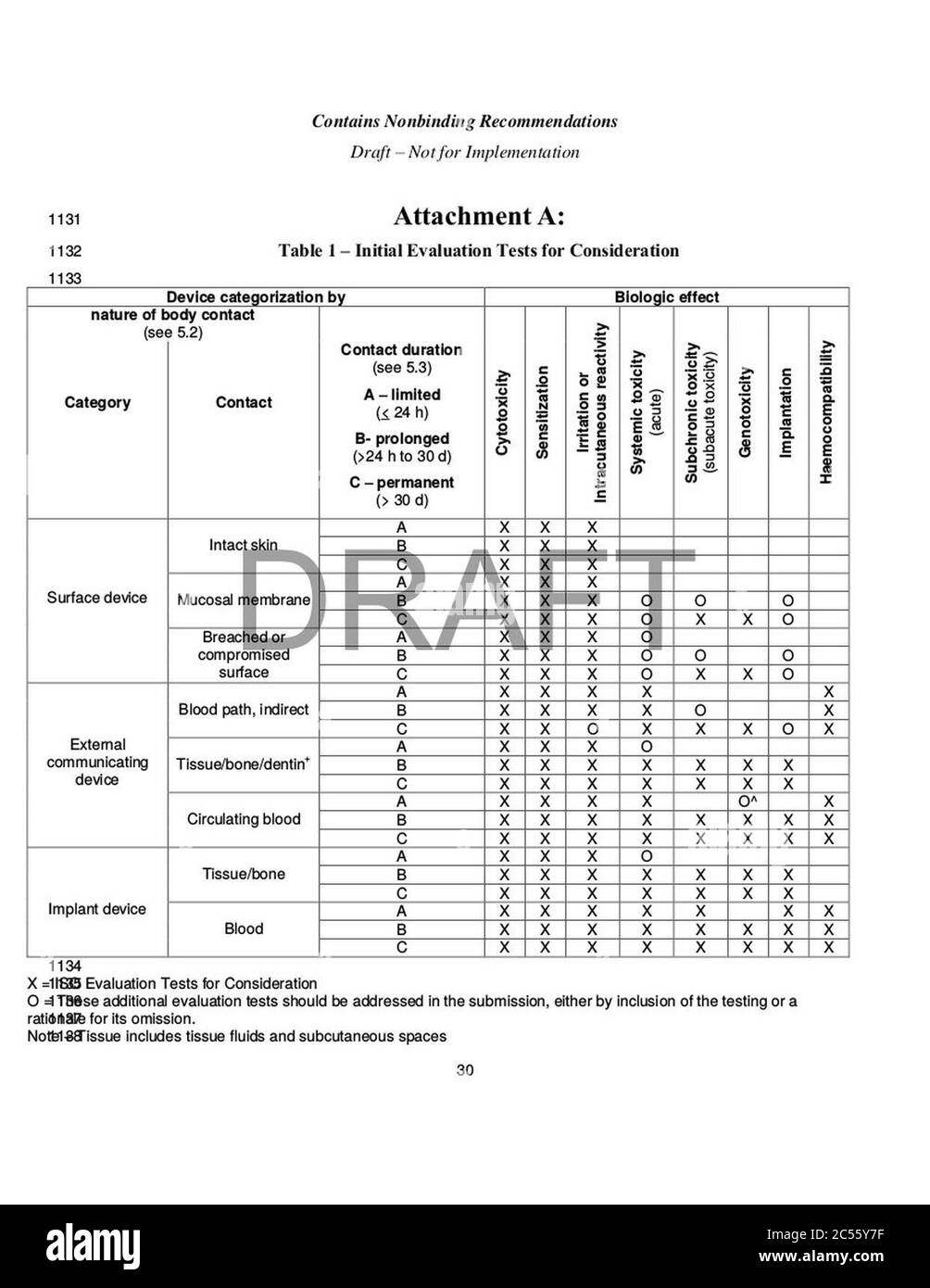
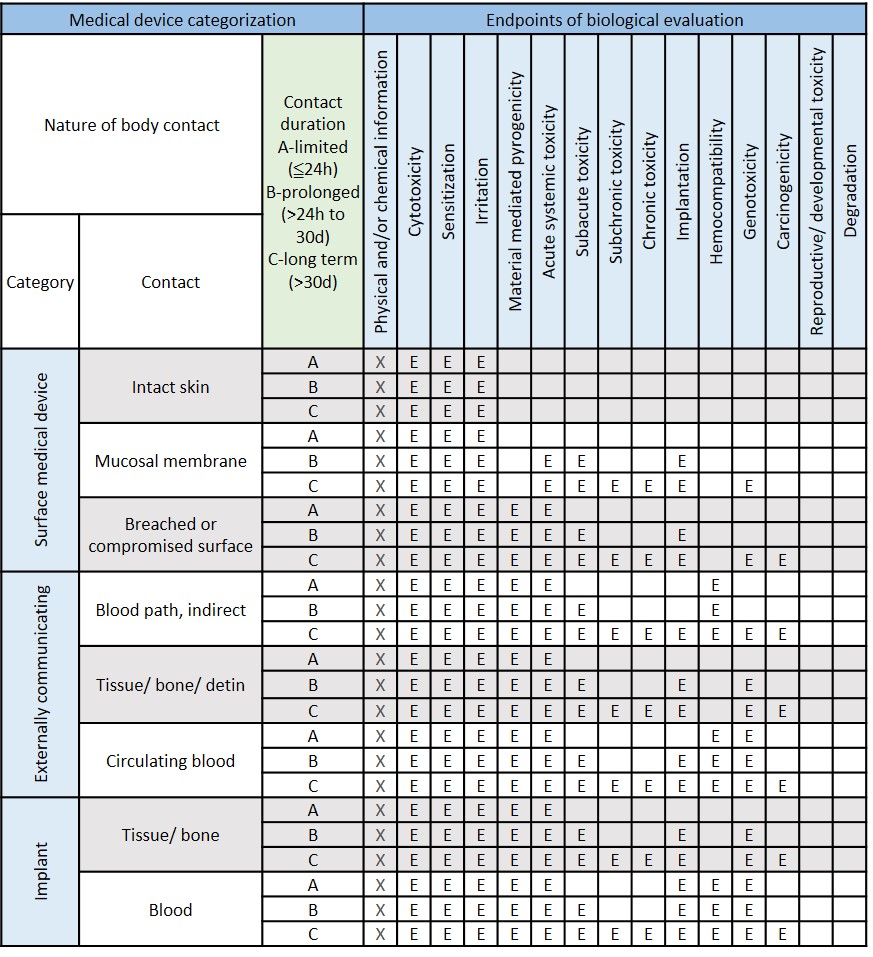
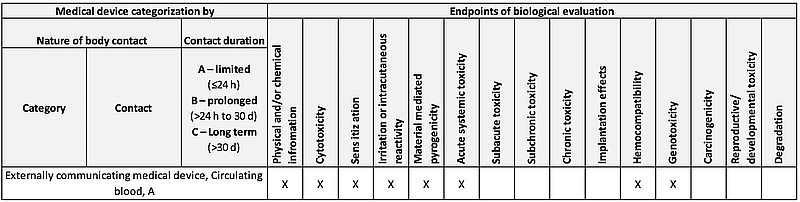
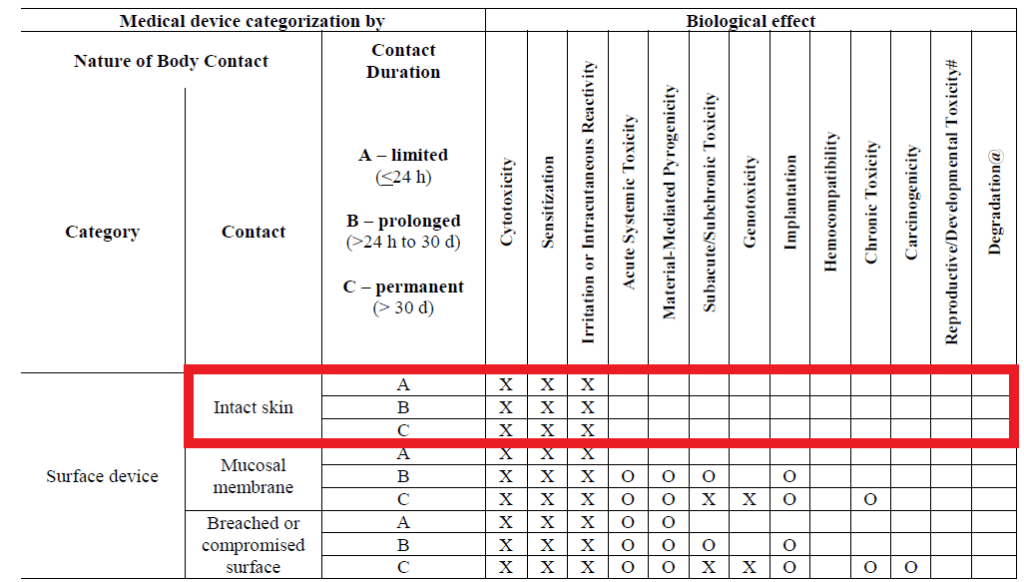
FDA FINAL GUIDANCE Use of International Standard ISO 10993-1, "Biological evaluation of medical devices - Part 1: Evaluation and testing within a risk management process" - Formiventos

Biological assessment of medical devices according to the ISO 10993-1 standard - GMED Medical Device Certification

Table I from Towards a logic-based extension of a relational software tool for coherent technical documentation of medical devices | Semantic Scholar

nueva edición norma ISO 10993-1:2018 biocompatibilidad con cambios relevantes | Red de Tecnologías Sanitarias y Productos Sanitarios