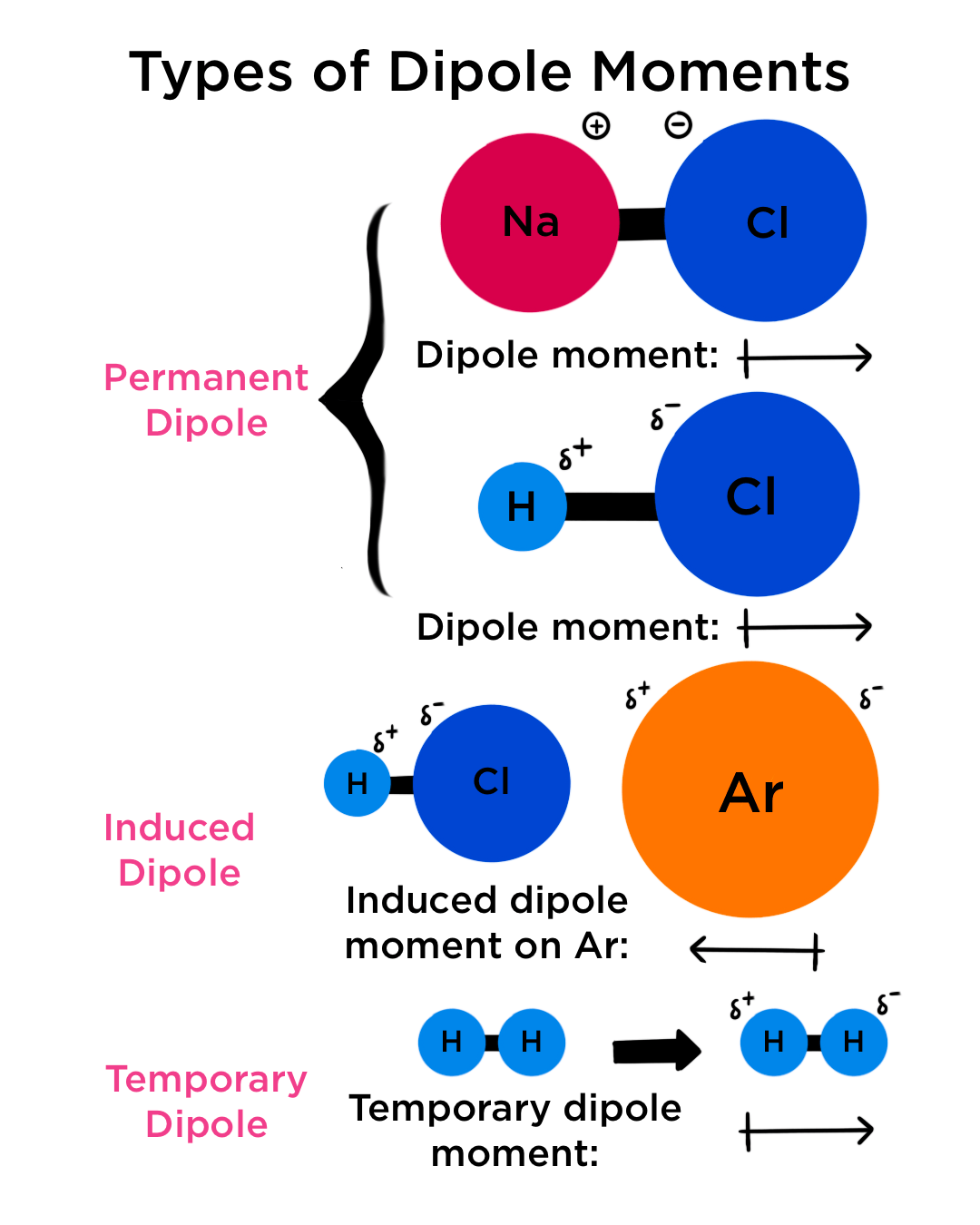
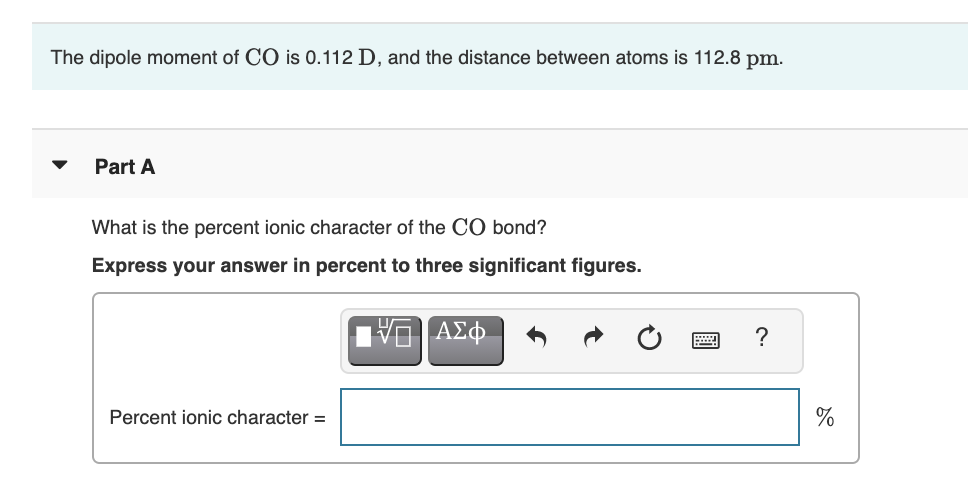
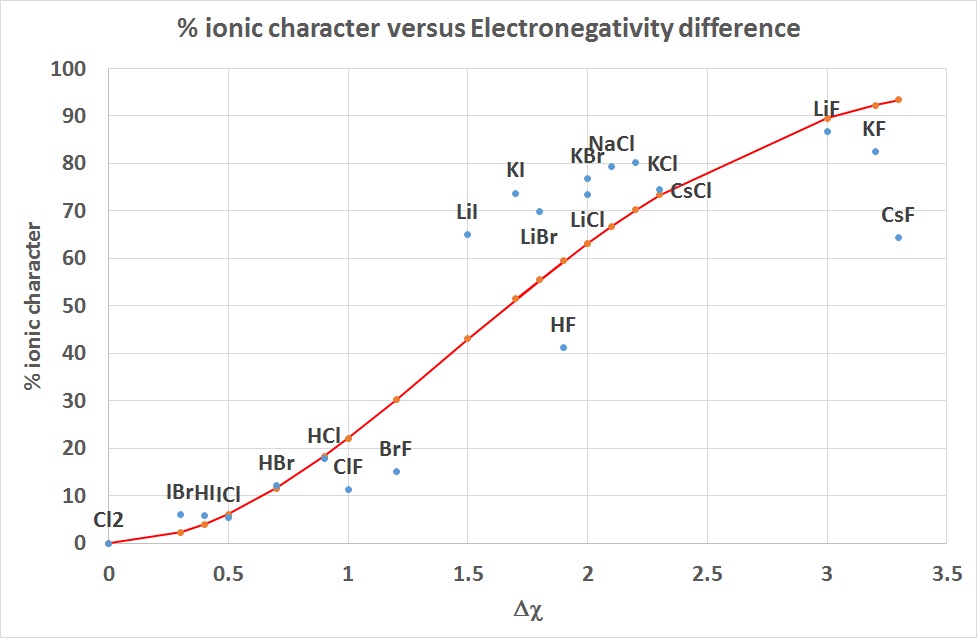
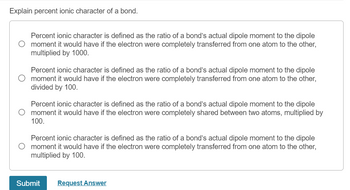
The experimental dipole moment of HCl is 1.03D and its bond length (distance) is 1.27 Å..... - YouTube
If the bond length and dipole moment of a diatomic molecule are 1.25 Å and 1.0 D respectively, what is the percent ionic character of the bond?

The dipole moment of HBr is 2.60 times 10^{-30} Cm and the interatomic spacing is 1.41 dot { A } . What is the percentage ionic character of HBr?

Ionic character in diatomic molecules. Bond ionic character (IC) for... | Download Scientific Diagram
Ionic 2: Module 'moment' has no exported member 'UnitOfTime'. · Issue #112 · urish/ngx-moment · GitHub
If dipole moment of HI is 0.38 D and bond lengthof HI is 1.6 A, then percentage covalent characterin HI bond is(1) 4.95

The observed dipole moment of AB is 1.2D and bond length is 1.2˚A. Then the percentage of ionic character is