Effectiveness of homologous/heterologous booster COVID-19 vaccination schedules against severe illness in general population and clinical subgroups in three European countries - ScienceDirect

Safety, immunogenicity, and efficacy of the mRNA vaccine CS-2034 as a heterologous booster versus homologous booster with BBIBP-CorV in adults aged ≥18 years: a randomised, double-blind, phase 2b trial - The Lancet

Immunogenicity and safety of homologous and heterologous booster vaccination of ChAdOx1 nCoV-19 (COVISHIELD™) and BBV152 (COVAXIN®): a non-inferiority phase 4, participant and observer-blinded, randomised study - The Lancet Regional Health - Southeast
Heterologous booster schedules are associated with increased protection against severe, omicron related COVID-19 outcomes
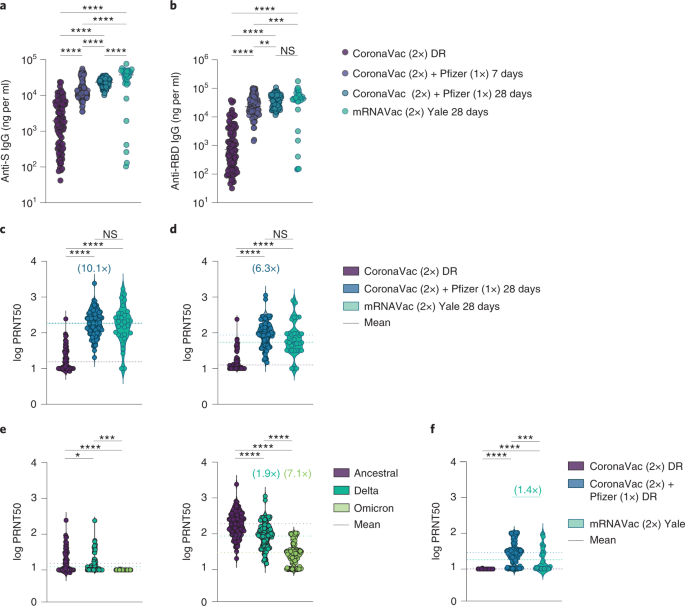
Neutralizing antibodies against the SARS-CoV-2 Delta and Omicron variants following heterologous CoronaVac plus BNT162b2 booster vaccination | Nature Medicine
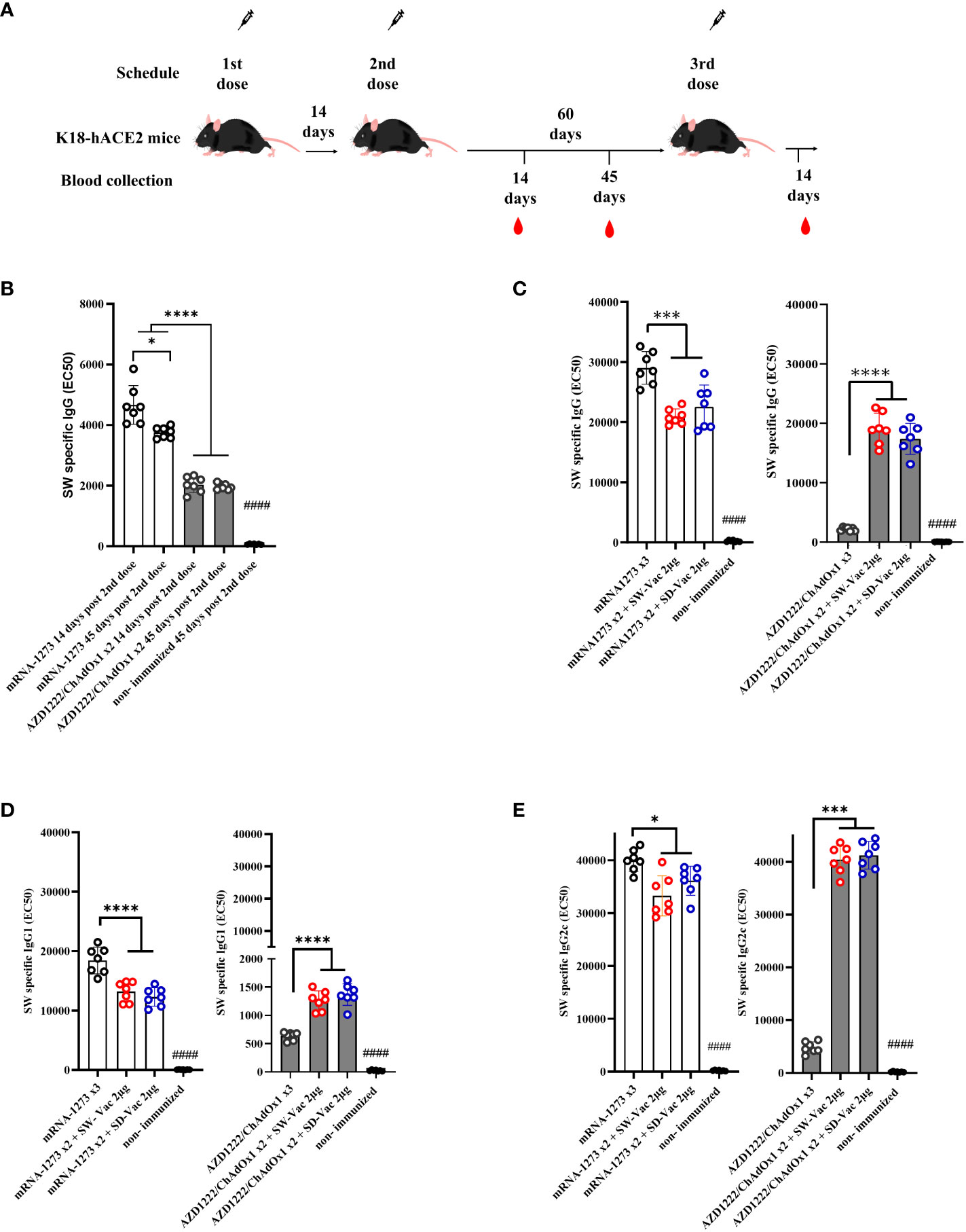
Frontiers | Heterologous booster with a novel formulation containing glycosylated trimeric S protein is effective against Omicron

iNCOVACC World's first Intranasal Vaccine to receive both Primary series & Heterologous booster approval | DD News
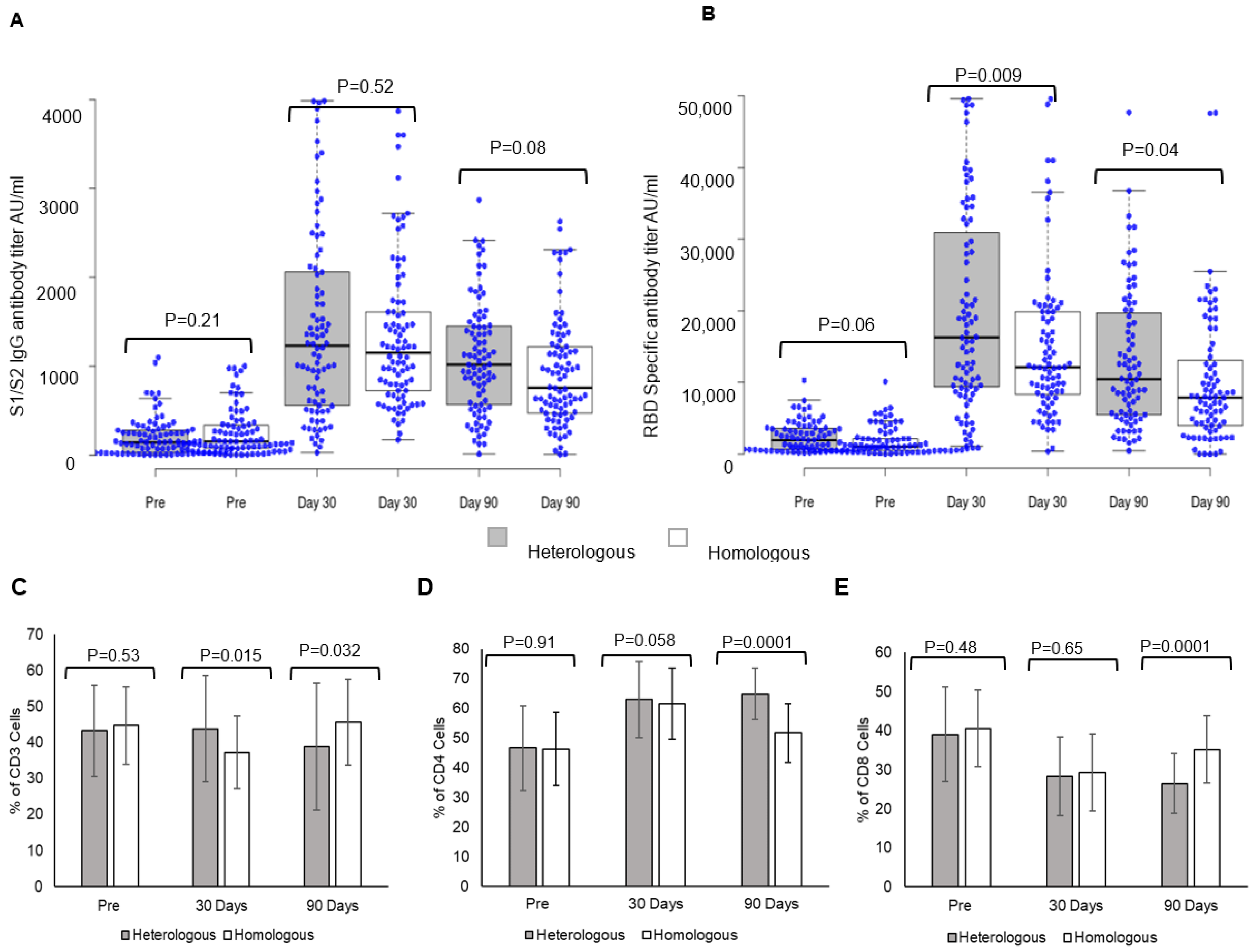
Vaccines | Free Full-Text | Heterologous Booster Dose with CORBEVAX following Primary Vaccination with COVISHIELD Enhances Protection against SARS-CoV-2

Vaccines | Free Full-Text | Heterologous Booster Dose with CORBEVAX following Primary Vaccination with COVISHIELD Enhances Protection against SARS-CoV-2

Comparative effectiveness of heterologous third dose vaccine schedules against severe covid-19 during omicron predominance in Nordic countries: population based cohort analyses | The BMJ
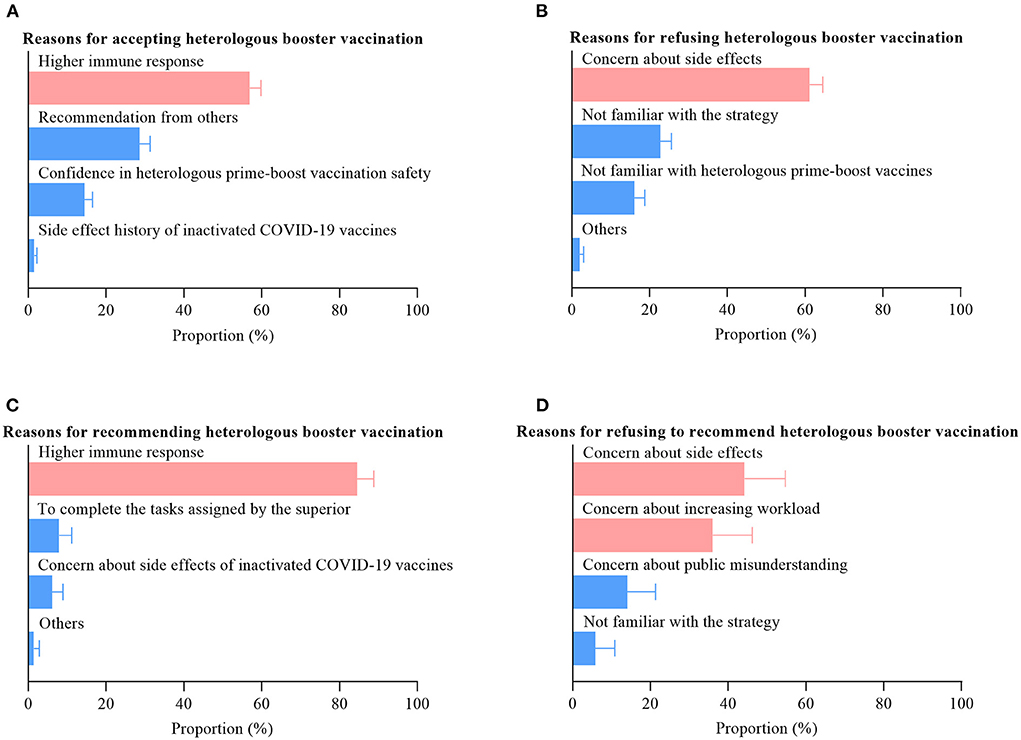
Frontiers | The acceptance to heterologous booster vaccination of COVID-19 vaccine among HCWs and targeted population: A cross-sectional study in central China

Safety and immunogenicity of heterologous versus homologous prime-boost schedules with an adenoviral vectored and mRNA COVID-19 vaccine (Com-COV): a single-blind, randomised, non-inferiority trial - The Lancet

Safety of heterologous primary and booster schedules with ChAdOx1-S and BNT162b2 or mRNA-1273 vaccines: nationwide cohort study | The BMJ

Heterologous versus homologous COVID-19 booster vaccination in previous recipients of two doses of CoronaVac COVID-19 vaccine in Brazil (RHH-001): a phase 4, non-inferiority, single blind, randomised study - The Lancet

Unravelling the enhanced vaccine immunity by heterologous KCONVAC/Ad5-nCoV COVID-19 vaccination | Signal Transduction and Targeted Therapy
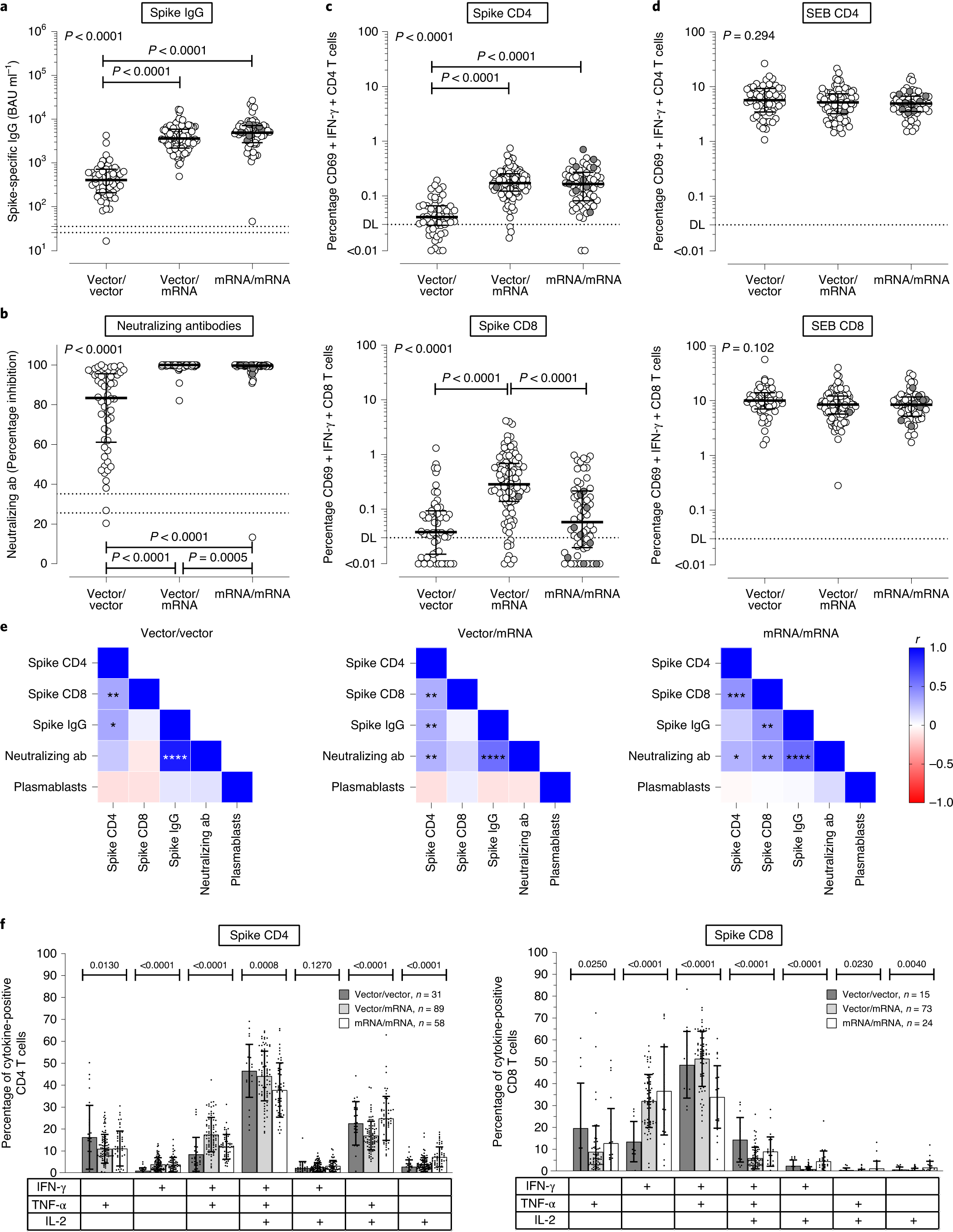
Immunogenicity and reactogenicity of heterologous ChAdOx1 nCoV-19/mRNA vaccination | Nature Medicine

Heterologous prime–boost vaccination with ChAdOx1 nCoV-19 and BNT162b2 - The Lancet Infectious Diseases