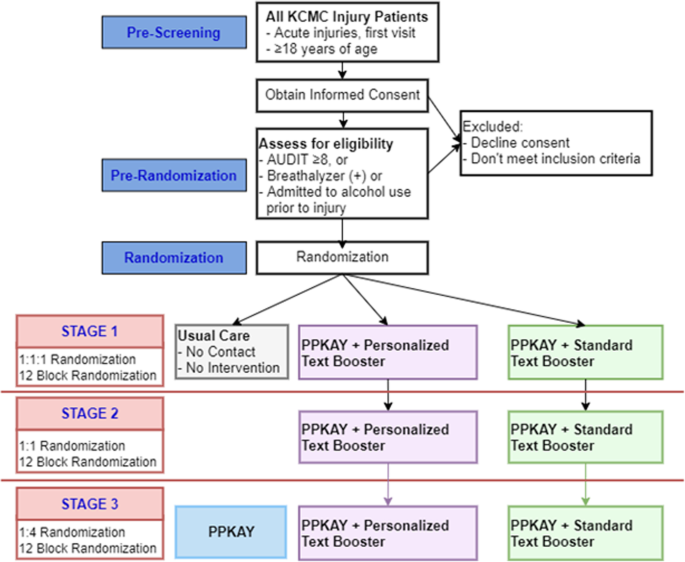
Efficacy and safety of a booster dose of influenza vaccination in solid organ transplant recipients, TRANSGRIPE 1-2: study protocol for a multicenter, randomized, controlled clinical trial | Trials | Full Text

Immunogenicity and safety of NVSI-06-07 as a heterologous booster after priming with BBIBP-CorV: a phase 2 trial | Signal Transduction and Targeted Therapy

Safety and immunogenicity of a bivalent SARS-CoV-2 protein booster vaccine, SCTV01C, in adults previously vaccinated with mRNA vaccine: a randomized, double-blind, placebo-controlled phase 1/2 clinical trial - eBioMedicine

Efficacy and safety of RTS,S/AS01 malaria vaccine with or without a booster dose in infants and children in Africa: final results of a phase 3, individually randomised, controlled trial - The Lancet

Cohort Profile:The Danish National Cohort Study of Effectiveness and Safety of SARS-CoV-2 vaccines (ENFORCE) | BMJ Open
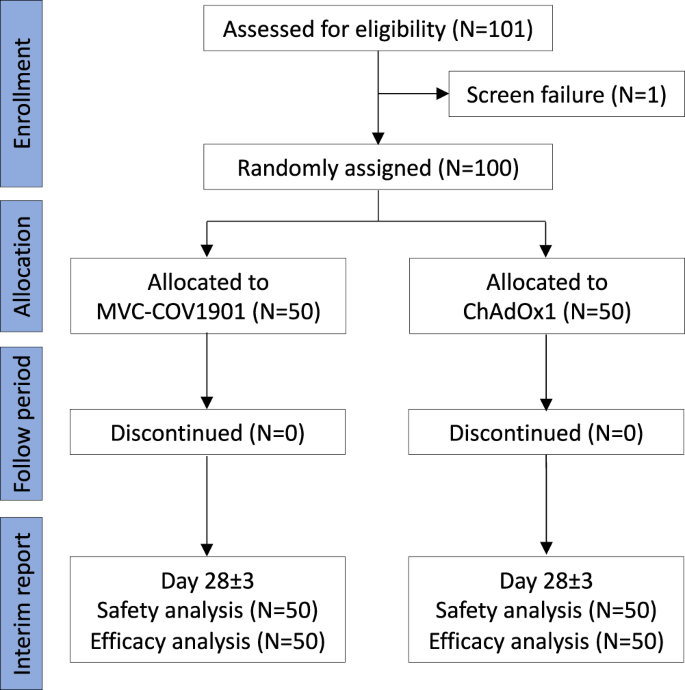
A randomized controlled trial of heterologous ChAdOx1 nCoV-19 and recombinant subunit vaccine MVC-COV1901 against COVID-19 | Nature Communications

Safety and immunogenicity of seven COVID-19 vaccines as a third dose ( booster) following two doses of ChAdOx1 nCov-19 or BNT162b2 in the UK (COV- BOOST): a blinded, multicentre, randomised, controlled, phase 2 trial -

Effects of temporarily suspending low-dose methotrexate treatment for 2 weeks after SARS-CoV-2 vaccine booster on vaccine response in immunosuppressed adults with inflammatory conditions: protocol for a multicentre randomised controlled trial and ...
NIH begins clinical trial evaluating second COVID-19 booster shots in adults | National Institutes of Health (NIH)
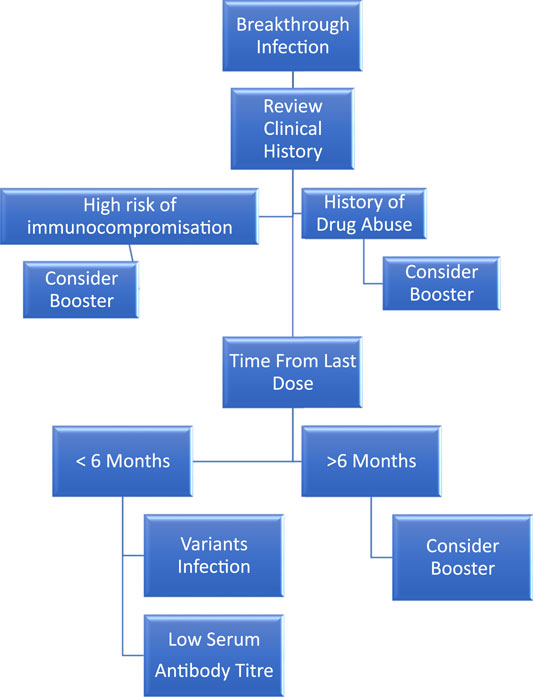
Frontiers | Review of Clinical Trials of COVID-19 Vaccination Booster in SARS-CoV-2 Variants Era: To Take It or Not To Take It

Immunogenicity and reactogenicity of BNT162b2 booster in ChAdOx1-S-primed participants (CombiVacS): a multicentre, open-label, randomised, controlled, phase 2 trial - The Lancet