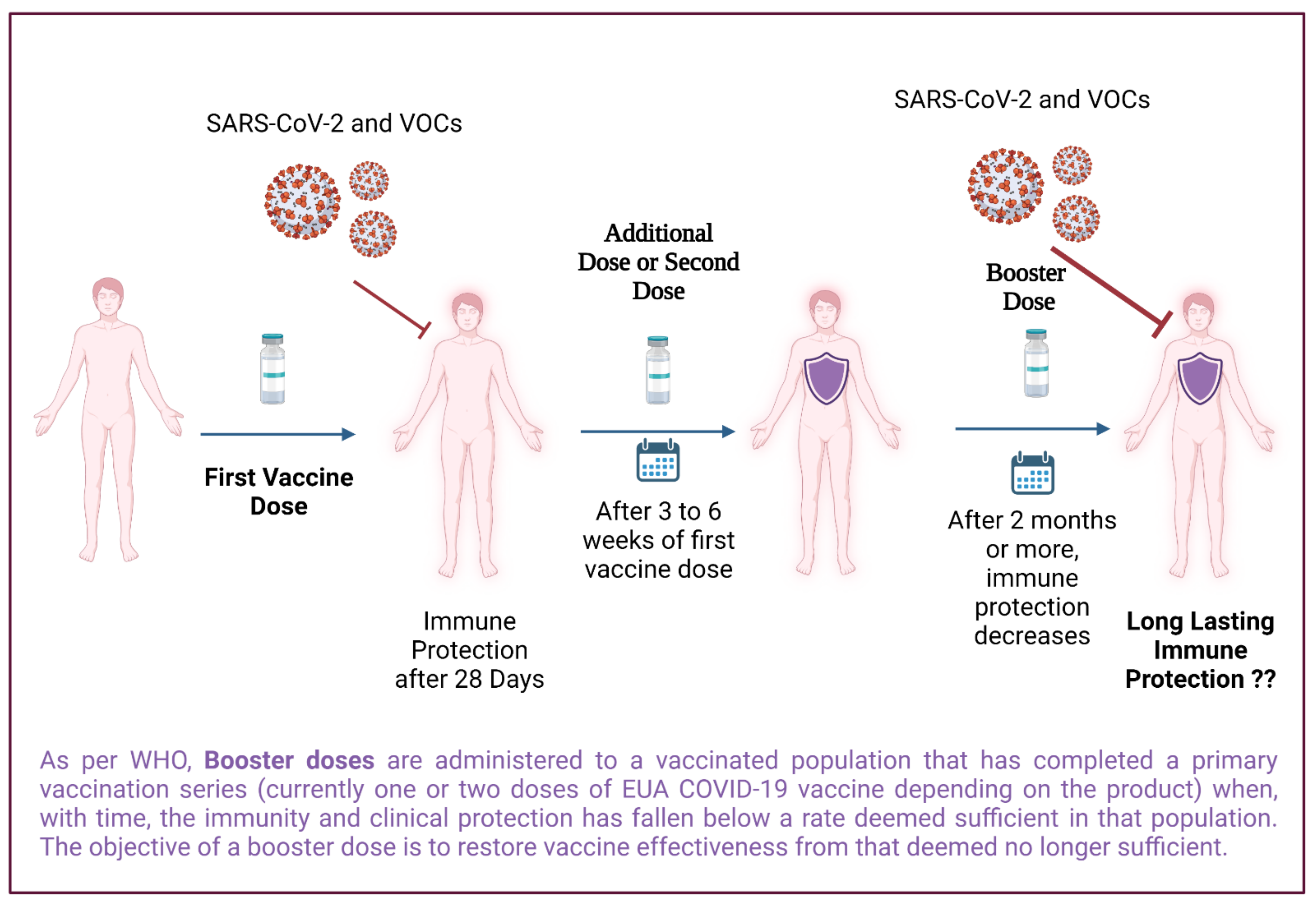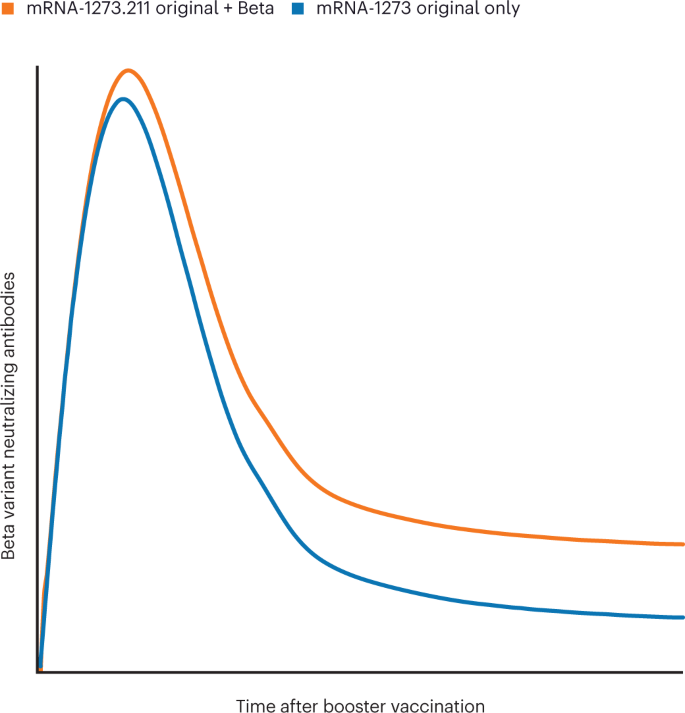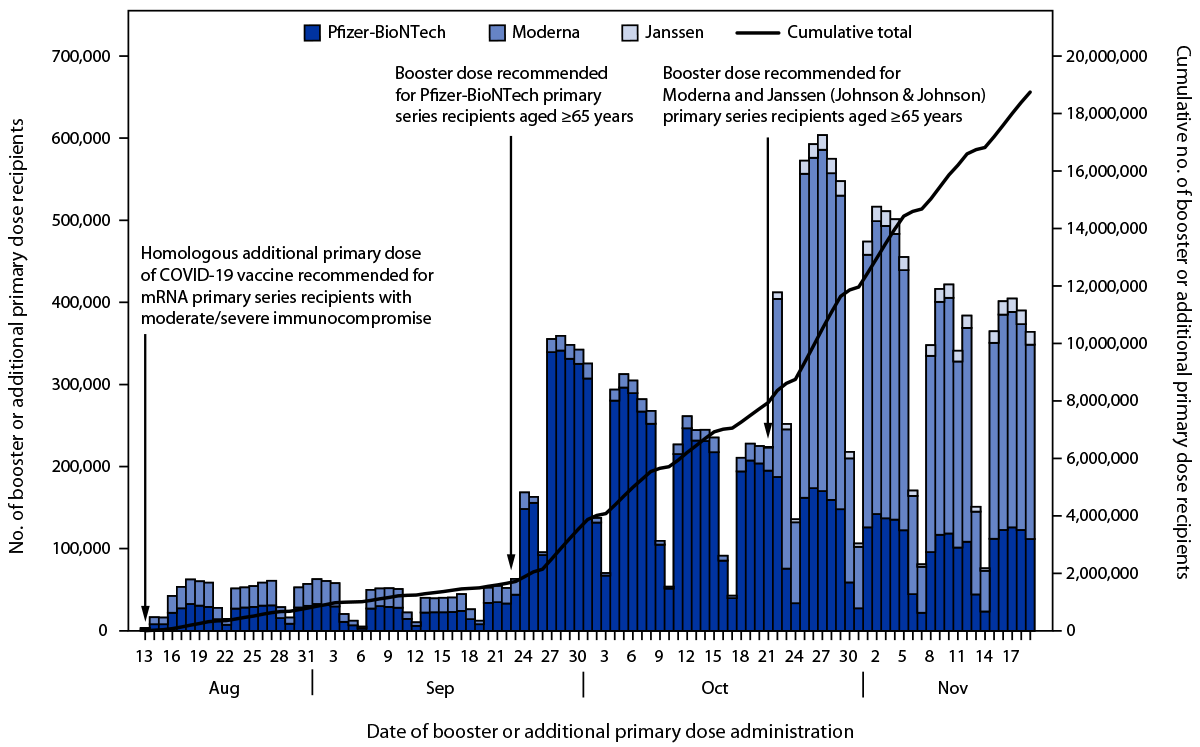
Booster and Additional Primary Dose COVID-19 Vaccinations Among Adults Aged ≥65 Years — United States, August 13, 2021–November 19, 2021 | MMWR
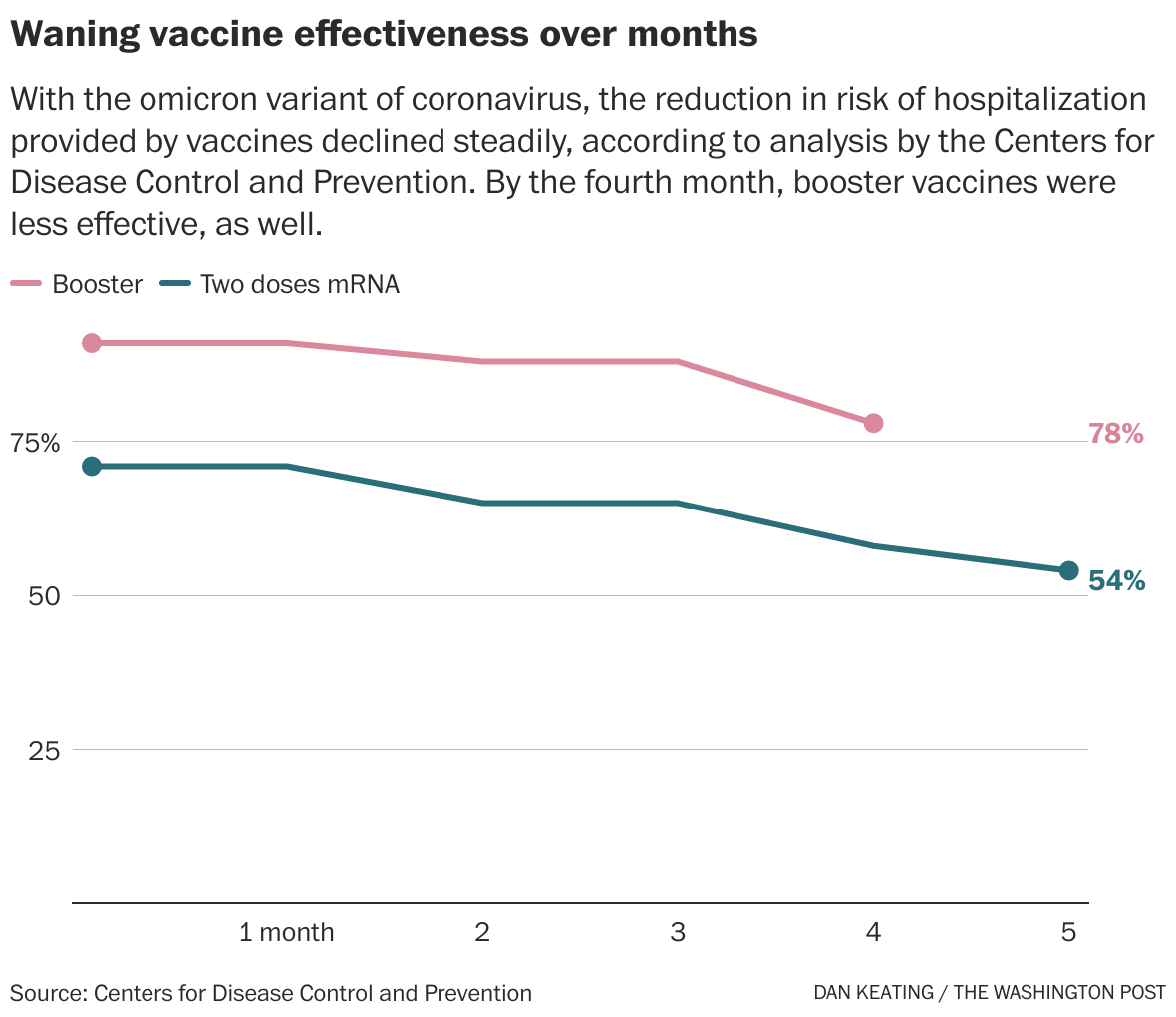
Boosters wane but showed protection against hospitalization during omicron, CDC study says - The Washington Post
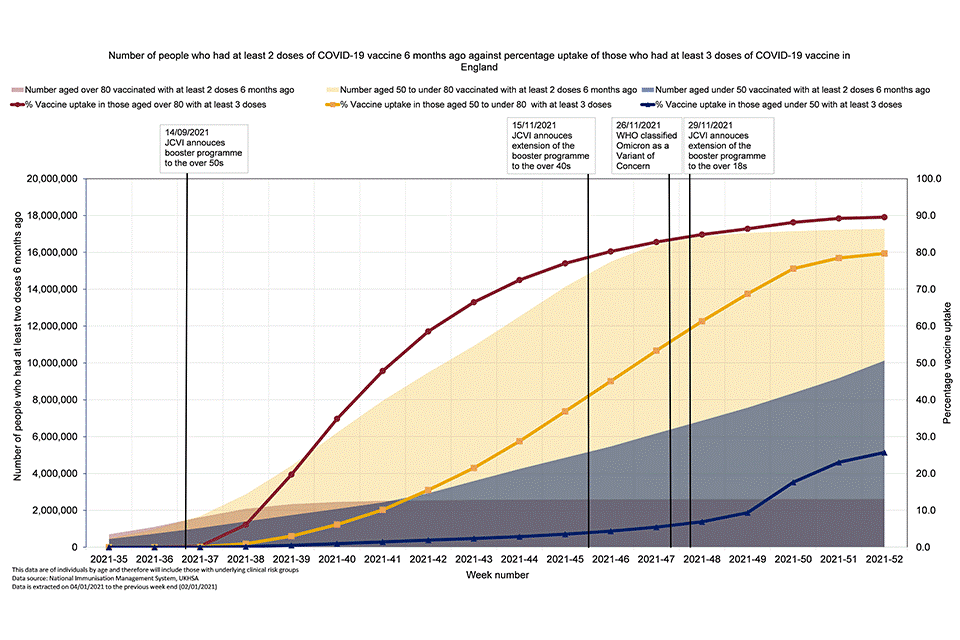
JCVI statement on the adult COVID-19 booster vaccination programme and the Omicron variant: 7 January 2022 - GOV.UK

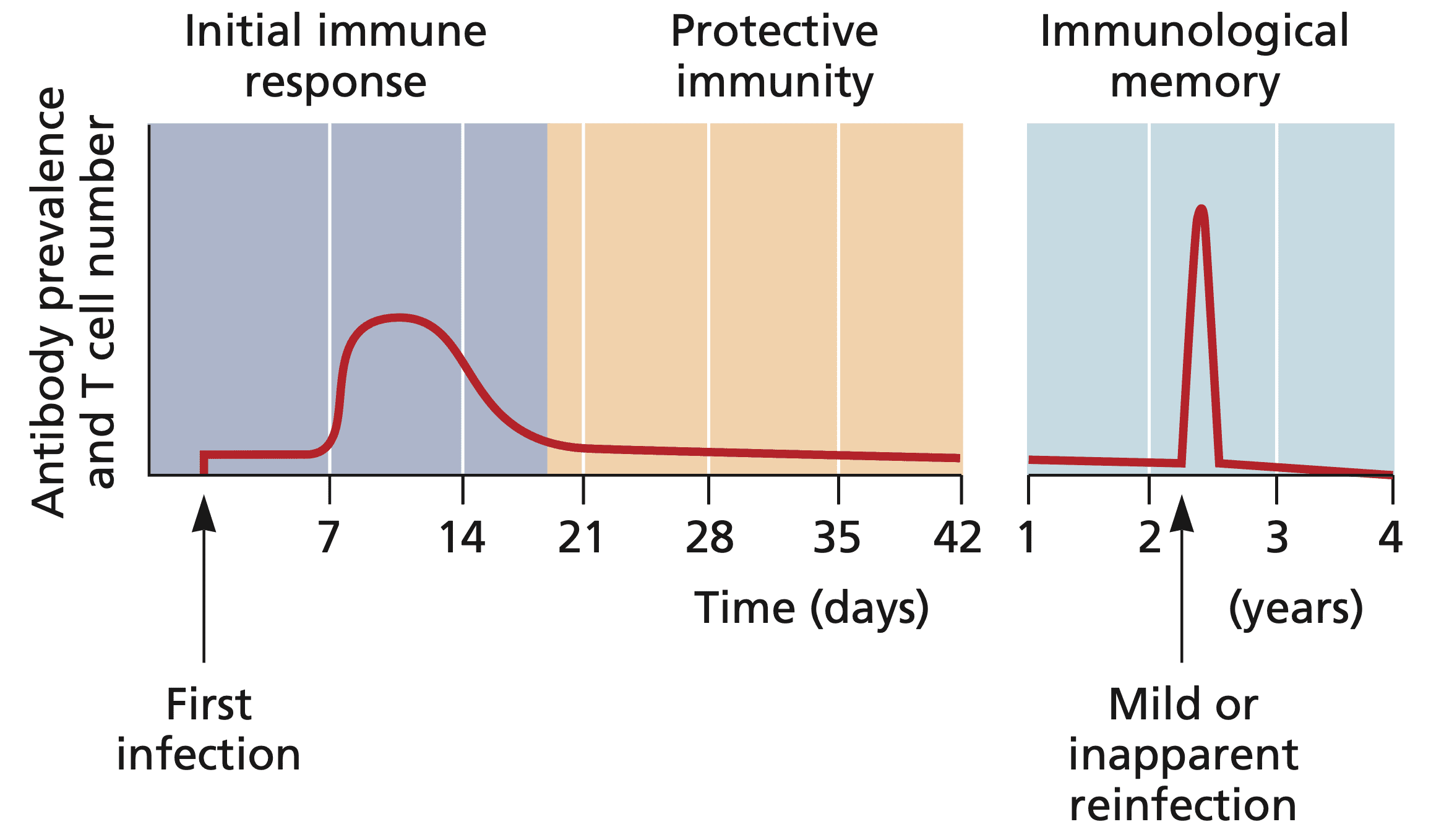
COVID-19 vaccine waning and effectiveness and side-effects of boosters: a prospective community study from the ZOE COVID Study - The Lancet Infectious Diseases

Comparative effectiveness of bivalent BA.4-5 and BA.1 mRNA booster vaccines among adults aged ≥50 years in Nordic countries: nationwide cohort study | The BMJ

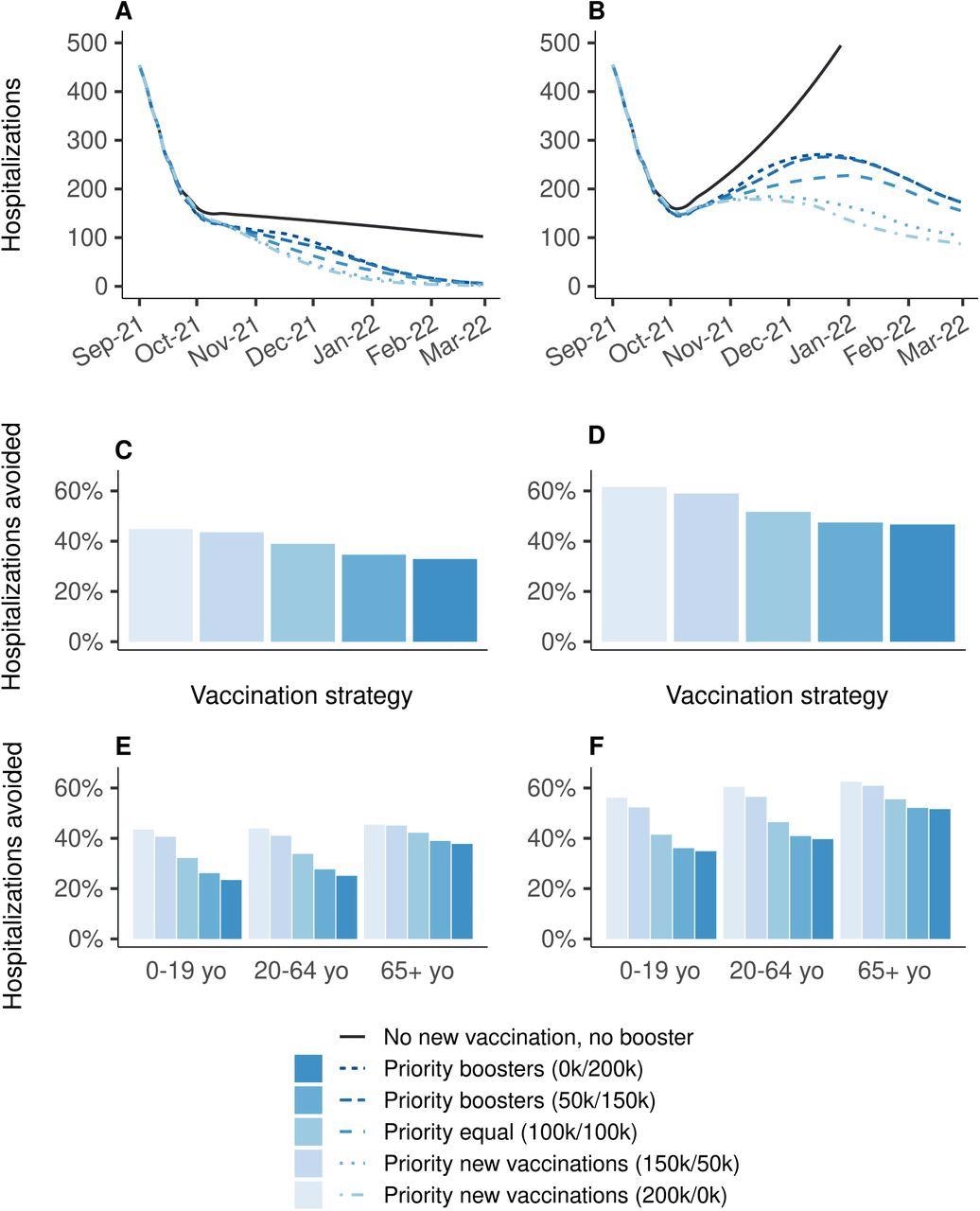
Comparative vaccine effectiveness against severe COVID-19 over time in US hospital administrative data: a case-control study - The Lancet Respiratory Medicine

Effectiveness of a third dose of the BNT162b2 mRNA COVID-19 vaccine for preventing severe outcomes in Israel: an observational study - The Lancet

Severity of omicron variant of concern and effectiveness of vaccine boosters against symptomatic disease in Scotland (EAVE II): a national cohort study with nested test-negative design - The Lancet Infectious Diseases
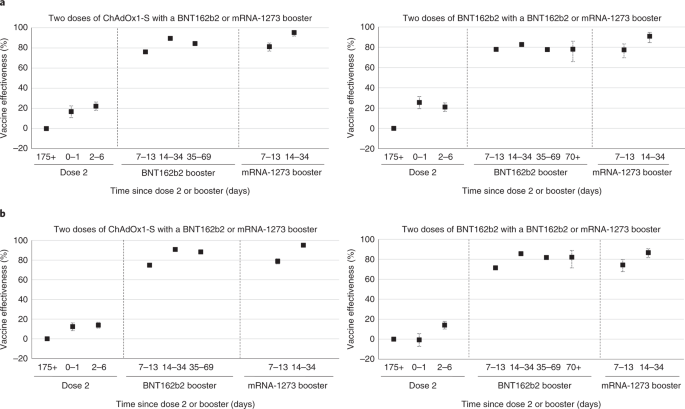
Effectiveness of COVID-19 booster vaccines against COVID-19-related symptoms, hospitalization and death in England | Nature Medicine

Effectiveness of primary series and booster vaccination against SARS-CoV-2 infection and hospitalisation among adolescents aged 12–17 years in Singapore: a national cohort study - The Lancet Infectious Diseases
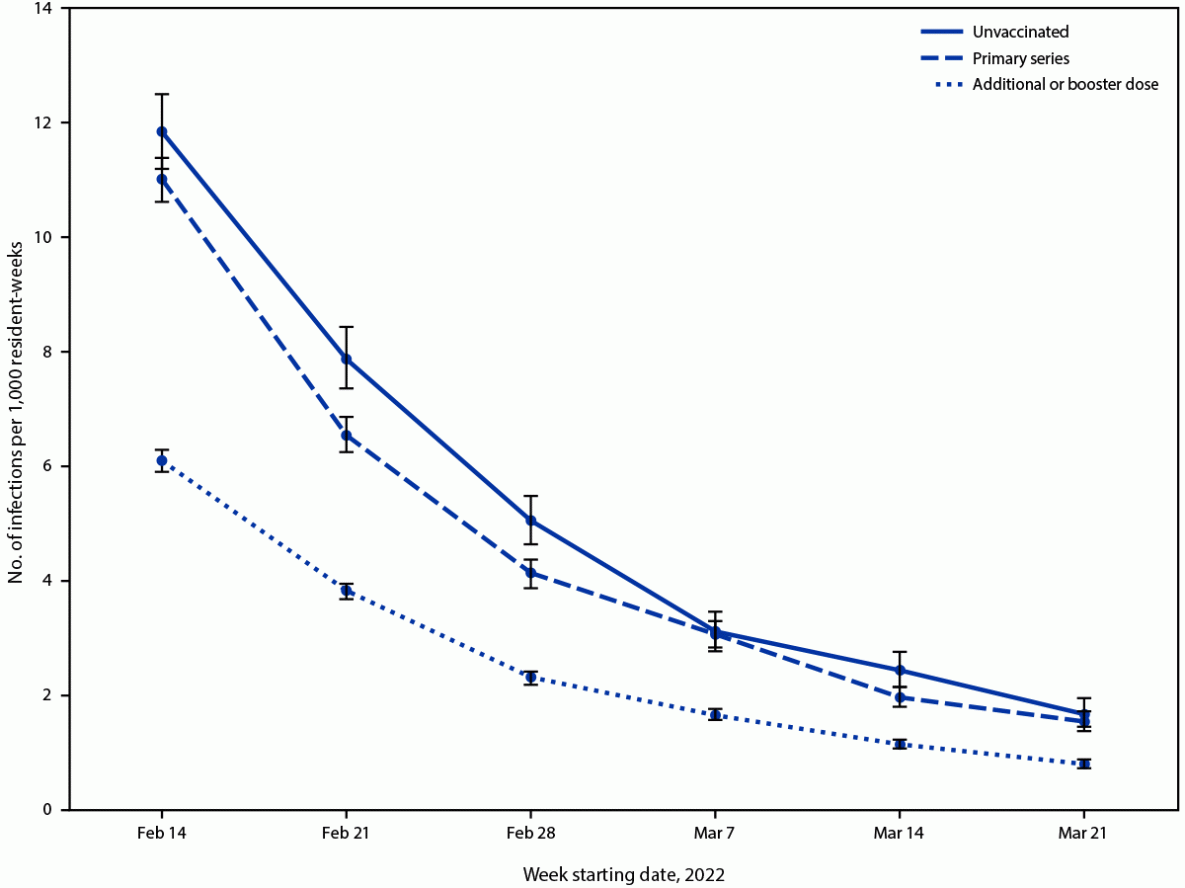
Effectiveness of a COVID-19 Additional Primary or Booster Vaccine Dose in Preventing SARS-CoV-2 Infection Among Nursing Home Residents During Widespread Circulation of the Omicron Variant — United States, February 14–March 27, 2022 | MMWR