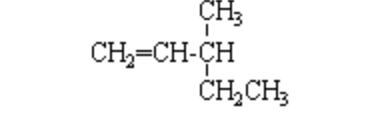1E)-1-methoxy-4-methyl-1-pentene - C7H14O, density, melting point, boiling point, structural formula, synthesis
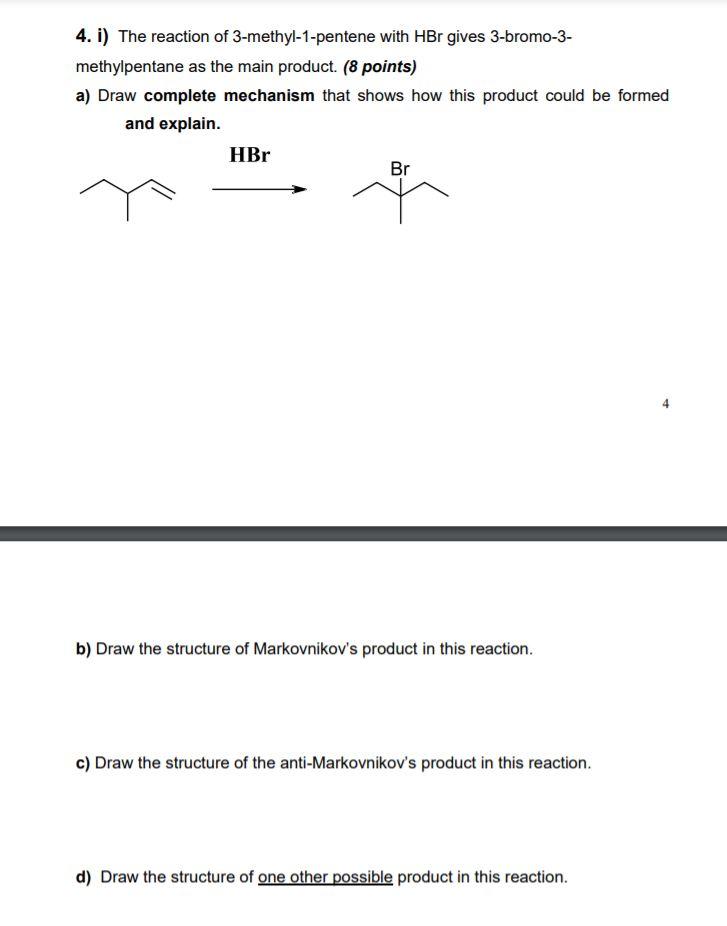
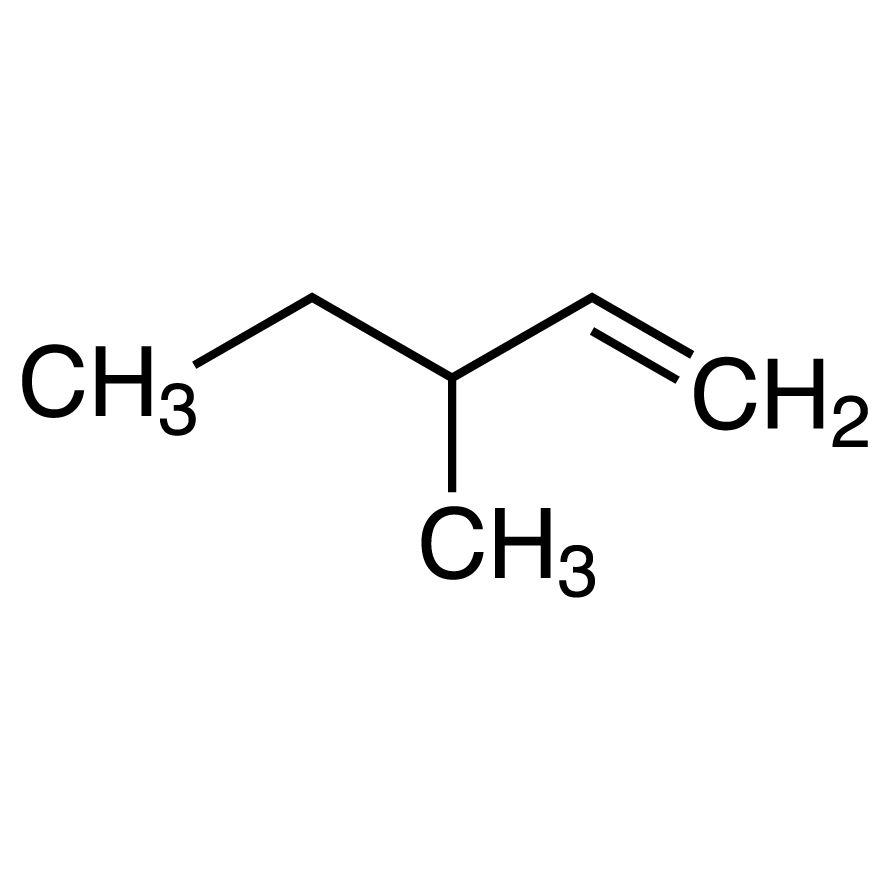
If a pure enantiomer of 3-methyl-1-pentene is treated with boiling solution of dilute H2SO4, how many different alcohols are expected in principle?Correct answer is '3'. Can you explain this answer? - EduRev
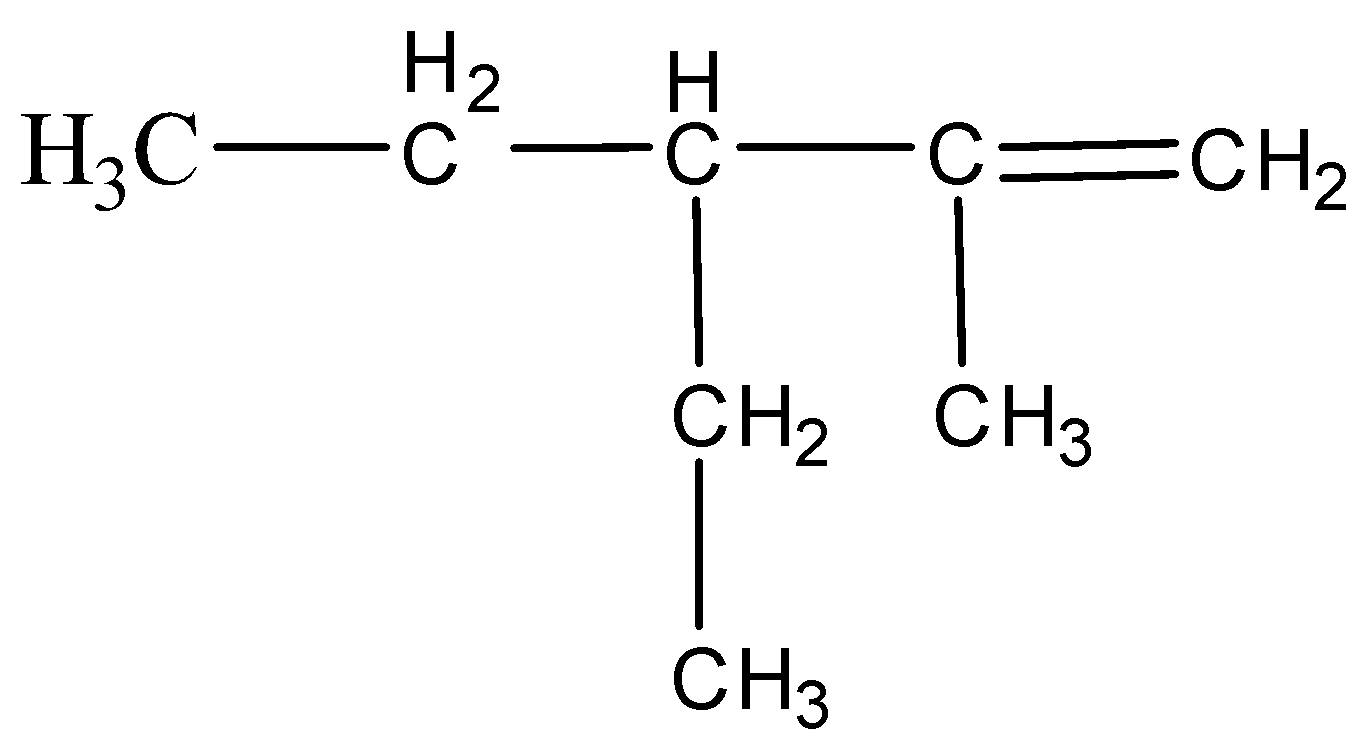
Name of the compound is:\n \n \n \n \n A.2-methyl-3-ethyl-1-penteneB.3 -ethyl-4-methyl-4-penteneC.3-ethyl-2-methyl-1-penteneD.3-methyl-2-ethyl-1- pentene